Copyright © All rights reserved. Made by Serif. Terms of use | Privacy policy
Tamar Estuary







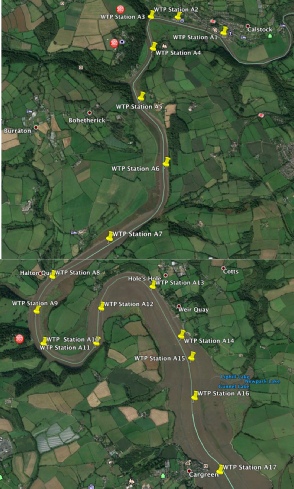
(Above) Map of stations sampled by Winnie The Pooh


(Above) Map of stations sampled by Falcon Spirit
(Above) Map of Plymouth area showing Saltash (Pontoon area)
(Above) Photo gallery of microscope images
Methods
The River Tamar was sampled via boat and at a stationary point on Saltash pontoon
04/07/18
Pontoon
Start Time: 9:00 UTC
End Time: 14:30 UTC
Location(50024’33”N, 004012’98”W).
Wind: 1-
Cloud cover: 6-
Samples at the pontoon were taken every 30mins to provide a time series. Temperature, salinity, oxygen, fluorescence and depth were measured using a T/S probe. Eularian methods were used to measure the flow speed and direction from the pontoon. This revealed the tidal cycle over the duration of sampling. A Sechii disk was used to estimate the light penetration and give an indication of the turbidity of the water column.
Niskin bottles were deployed on the hour to obtain water samples which were filtered
to gain chlorophyll a (chl-
Boat: RV Falcon Spirit
Start Time: 10:20 UTC
End time: 14:17 UTC
Wind: 2 (Beaufort Scale)
Cloud cover: 8/8
We sampled at 9 different stations covering a total of 8 different locations along
the area around Plymouth breakwater and up to Saltash. Past this point, the smaller
vessel named ‘Winnie the Pooh’ would sample the more narrow stretches of the upper
estuary. At each station, time , location and weather was noted and a CTD rosette
containing 6 Niskin bottles was deployed. Smaller samples were extracted from the
trapped water in the Niskin bottles . Samples were filtered for chl-
A secchii disk was then used to measure the sub-
Boat: Winnie The Pooh
Start Time: 9:00 UTC.
End Time: 14:30 UTC
Wind: 1 (Beaufort Scale)
Cloud cover: 8/8
We travelled up the Tamar estuary until reaching Calstock where surveying began. The same measurements were taken on the Winnie the Pooh as on the RV Falcon Spirit. A total of 18 samples were collected at intervals between Calstock and Cargreen towards the mouth of the estuary.
Depth was sampled using an on-
Zooplankton abundance sampled using a zooplankton net at 4 different stations (A1, A6, A12 and B18). The revolutions were noted before and after the sample. The net was washed and 10% formalin was added to preserve the samples before analysis. A sample of zooplankton was also collected using a filter syringe and added to a brown glass containing lugols.
Physical
Light Attenuation
Light attenuation and the depths within the water column at which only 1% of the surface irradiance is able to penetrate (the 1% light depth) were calculated from the Secchi disc depths measured at each station along the lower estuary. The light attenuation coefficient (k) was highest at Stations 3(B), 4(C), and 8(H); and lowest at Station 9(I). In agreement with this, the 1% light depth was shallowest at Stations 3,4 and 8; and deepest at Station 9. Station 9 was the most seaward of the sampling stations, while Stations 3, 4, and 8 are located in the vicinity of riverine inputs to the estuary. River water often contains higher sediment and organic loads than seawater, and these particles cause scattering and/or absorption of incident light. It is therefore likely that it is this potential difference in particulate load within the water column which resulted in the observed differences in light attenuation at these points. This is significant for the biology of the area as the 1% light depth is generally accepted as representing the base of the euphotic zone, and this is the area within the water column where there is enough light available for photosynthesis to occur. It should however be noted that the Secchi disc measurements were taken at different times throughout the day, and therefore other factors such as tidal state and weather may have had an effect on the data gathered.
The Secchi disc depth measurements collected as part of the time series data from Saltash Pontoon were used to calculate the light attenuation coefficient (k) and 1% light depths at different times throughout the day. Within the sampling period k fluctuated, however it reached its lowest value at 10:05 UTC, and its highest value at 15:30. The values for the 1% light depth correspond to these values, being shallowest at 15:30 and deepest at 10:05. Increases in light attenuation can result from high abundances of phytoplankton in surface waters, as they absorb and scatter some of the incident light. As some phytoplankton groups are known to undertake diel migrations in response changing light levels (Olli, 1999), this may be a factor underlying this observed trend, with phytoplankton moving towards the surface throughout the sampling period, thus increasing light attenuation. Further data backing this up is the observed increase in chlorophyll concentration within the water column throughout the sampling period
Richardson’s Number
Above are the Richardson’s number calculated from the data collected from the Tamar estuary. Only 4 stations had sufficient data to produce these results. A Richardson’s number of <0.25 shows that the water at this depth can be classified as turbulent.
Chemical
Dissolved Oxygen Measurements
Samples taken on the falcon spirit had 1ml manganous chloride and 1ml of alkaline iodide added to them immediately after collection.
Oxygen was captured in the precipitate. To release the O2, 1ml of sulphuric acid (H2SO4) was added. A magnetic stirrer was added and the bottle was placed into the bottle which was then placed into an incubator containing seawater. The sample was stirred until the precipitate was completely dissolved.
An auto titrator was used to add sodium thiosulfate (Na2S2O2 x H2O) (0.22 normality)
until the sample became clear. Clarity was determined with an end-
Phosphate Analysis
10ml of filtered sample collected from the estuary were put into numbered sample bottles. This included a repeat every five samples.
Standards and blanks were then prepared for comparison and calibration by making solutions of a known concentration and measuring them in the same spectrophotometer.
Mixed reducing reagent (MRR) solution was prepared using a set ratio. 1ml of this was added to each sample. Samples were left for 1 hour to develop before being put into the spectrophotometer.
We encounted issues with the standards however as no phosphate had been added and thus the standards had to be redone before being put through the spectrophotometer. Once done, we recorded the absorbance for each standard and all the samples.
A Theoretical Dilution Line and Mixing diagram was then plotted using the phosphate concentrations
Silicon Analysis
Silicon standards were produced by diluting the stock solution into 9 tubes and 3 additional tubes which acted as blanks.
Collected samples from the estuary were then prepared by distributing the samples into polypropylene tubes before adding 2ml of molybdate reagent to each 5ml of sample.
Once mixed the samples stood for approximately 10 mins before adding a MRR (10 parts metal sulphite, 6 parts oxalic acid, 6 parts sulphuric acid + 8 parts MQ water). Samples were then left to develop for a further two hours to allow for full colour development.
The absorbance of the samples and standards were then measured in the spectrophotometer at a wavelength of 810nm. The data for the absorbance was then used to construct an estuarine mixing diagram.
Chlorophyll Concentration Analysis
The filters collected from the various estuary stations were used to quantify the amount of chlorophyll in the water column at different depths. The filters had been previously stored in acetone immediately after collection. To calculate the chlorophyll concentration, the following equation was used:
In the lab the filters were removed from their tubes and the remaining acetone was placed into the fluorometer. This was then used to produce a resistance reading. The volume of sea water and acetone used was kept constant throughout.
Mixing Diagrams and the theoretical dilution line (TDL) show how solutes act in the estuarine environments (See link below). The TDL is created by drawing a line through the points of lowest salinity (river water) and the point of highest salinity (seawater). The first graph shows silicon and how it behaves in the Tamar estuary. The points drop below the TDL which shows a removal of dissolved silicate from the water column, the proposed explanation for this decrease in silicon is the blooming of phytoplankton such as diatoms which use to silicon to build shells. Phosphate, a crucial nutrient for protein and DNA production. Phosphate shows different mixing to silicon. Phosphate shows removal higher in the estuary and addition in the lower estuary. This change in mixing could be caused organisms taking up phosphate initially then later in the estuary when it reaches more industrialised zones, outflows may introduce further phosphate causing the addition of phosphate to the estuary. Nitrate, the final nutrient studied by us in the Tamar shows conservative mixing. None of the nitrate leaves or enters the system through processes in the water. Depth profiles for phosphate show that phosphate levels do not rise above 0.5 umol/L regardless of depth. It also shows that there are is a large variety of values at similar depths. Possibly relating to the differences in physical parameters in the stations up and down the estuary. Silicon values have a much larger variability in comparison to the phosphate numbers. This higher variability could be attributed to the fact that some of the phosphate is removed while some of it is added as evidenced in the mixing diagrams. This will accentuate the minimums and maximums of the graph as a result.
Biological
Zooplankton
10ml of sample was measured out. 5ml of this was then pipette into Bogorov dishes for analysis under the microscope.
Using an identification guide, the species found and their abundance were recorded.
Volume of seawater sampled was calculated first by calculating towing distance:
This was then substituted into the following formula to find the volume sampled:
Where:
Π= 3.14159
R=Radius of plankton net open (m)
L = Towing distance (m)
V= Volume of seawater sampled (m3)
The number of zooplankton per m3 of seawater was then calculated using: ![]()
Where
n= Number of zooplankton in 10ml of a 1l sample.
V=volume of seawater sampled (m3)
N= Number of zooplankton per m3 seawater
![]() Phytoplankton
Phytoplankton
10ml of SW sample were used and 1ml of Lugols solution was added to each sample per 1ml.
The SW and Lugols mixture was then placed in a settling tube to concentrate.
Liquid was poured off to obtain a concentrated sample for study under the microscope.
1 ml of the concentrated mix was extracted and studied under a light microscope.
Using an identification guide, we examined 20 x 5 columns giving us a total of 100 squares on the viewing slides.
29 different species were identified in the samples from the Falcon Spirit. The pie chart shows that the species that had the highest abundance were ciliates (14%), diatoms (31%)and dinoflagellates (14%), all other species had much lower abundances.
We found a much higher biodiversity in the samples collected from the falcon spirit, compared to the Winnie the Pooh. The species with the highest abundance found in the samples collected by the two boats are different. This could be due to the differences in the conditions in the areas sampled by the different boats. As the Winnie the Pooh collected data towards the river whereas the falcon sampled in the sound.
Zooplankton Results
Approximately 50% of the zooplankton sampled by the Falcon Spirit (mouth of the estuary) consisted of Cirripedia larvae. A relatively large proportion of the zooplankton is Copepod and Copepod nauplii, together consisting another ~45% of the population (seen in pie chart)
A smaller proportion is made of Cladocera, Echinoderm larvae. Among the less densely populated groups of zooplankton were; mysidacea, polychaeta larvae, gastropod larvae, hydromedusae and fish larvae.
Zooplankton collected from further up the estuary mainly consisted of copepods which contributed over 75% of the population. The other 20% of the zooplankton population was made of approximately even proportions of other groups such as copepod Nauplii, polychaete larvae and Cladocera. In contrast with the samples from the Falcon Spirit, Cirripedia larvae were absent from the water column.
A larger proportion of copepod Nauplii were found at station 8 and 18 than at stations 2, 9, 1 and 6 which mainly consisted of Copepod (seen bar chart). At station 18 around 20% of the zooplankton consisted of Siphonophorae. These changes reflect the stenohaline tolerances of the various groups as was found by Froneman (2004) where zooplankton community structure was primarily controlled by the presence/absence of the marine environment and freshwater was predominantly copepods in biomass. Gunter (1961) noted that a correlation between salinity and size existed. Larger organisms tend to be found further out towards the sea at higher salinity and often migrate downstream as they grow.
Overall the greatest zooplankton abundance was found at station 8 followed by station 2.
Phytoplankton Results
The dominant phytoplankton group sampled at the stations by the RV Falcon Spirit was Pleurosigma spp – accounting for around one third of the total community structure. Cosinodiscus spp. and Ciliates individually contribute another 20% of the community.
The phytoplankton community sampled further to the head of the estuary was comprised of over 75% of Coscinodiscus spp. Coscinodiscus is present further out of the estuary at station 18 but seems absent at stations 8, 7 and 5. Station 1 on the other hand shows only a small percentage of Coscinodiscus and the majority of the community is comprised of Diatoms. Station 2 is comprised almost entirely of Coscinodisucs and Ceratium spp. Overall, plankton concentration was significantly higher at station 6 than all other stations by several times.
The biomass of plankton mid estuary is consistent with the nutrient mixing diagrams which show removal along the estuary. Abundance would also be expected to be greatest at the fresh/saltwater interface in the estuary as both freshwater and marine species can survive there. This is supported by the findings of Ahel (1996) who found that the maximum concentration and carotenoid pigments were found in the brackish area of the highly stratified Krka estuary.