
The aim of this survey was to create a benthic habitat map for the area between Carricknath
Point and Shag Rock. This was done by using sidescan sonar in conjunction with ground-
Sidescan model: Kongsberg dual-
Operating frequency (kHz): 100 and 410
Paper trace software: GeoAcoustics
Navigation software: HYDROpro navigation
Trackplot software: Surfer
Co-
Sidescan swath (m): 150
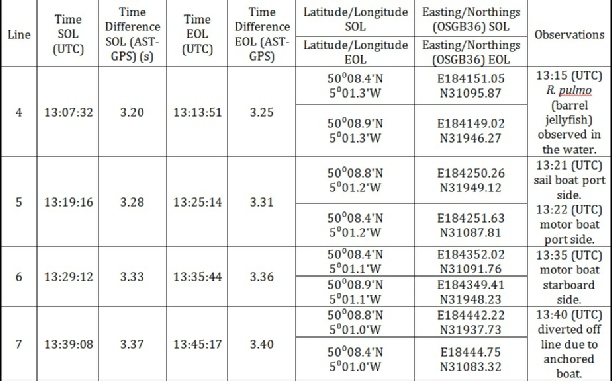
In total, four transects were taken and numbered Lines 4-
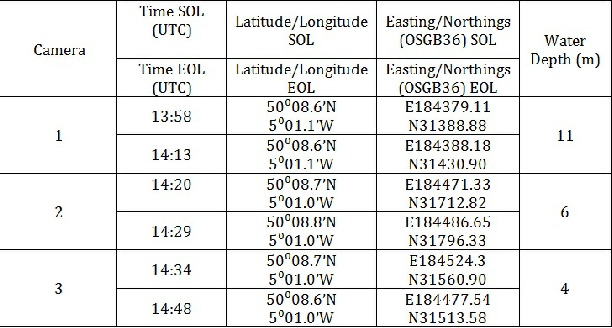
Three camera transects were taken by selecting a position within the sidescan transects and allowing the boat to drift for an average of 12 minutes. This gave a curved transect which can be seen on the benthic habitat map. The camera used was a boat tech camera. Some issues were encountered on camera transect 1 due to a fuzzy picture, making identification difficult but not impossible. No grab was taken as we were located in an SAC.
Date: 23.06.2015
Time: 12:50 (UTC)
Location:
Line 4: Start of line: 50° 08.4’ N 5°01.3’ W
End of line: 50° 08.9’ N, 5° 01.3’ W
Line 5: Start of line: 50° 08.8’ N 5°01.2’ W
End of line: 50° 08.4’ N, 5° 01.2’ W
Line 6: Start of line: 50° 08.4’ N 5°01.1’ W
End of line: 50° 08.9’ N, 5° 01.1’ W
Line 7: Start of line: 50° 08.8’ N 5°01.0’ W
End of line: 50° 08.4’ N, 5° 01.0’ W
High tide: 9:20 4.3m
Low tide: 15:50 1.5m
Cloud cover: 5/8
Sea state: 2/10
Disclaimer-
Falmouth 2015 Group




Benthic Habitat Map – Results and Discussion
Click Here to view Habitat Mapping Poster
Click Here to view Map as PDF
Rock – Unknown Flora
This habitat type comprises of the areas enclosed by boundaries 5, 6 and 10. We have
no ground-
Muddy Substrate
We have inferred that boundaries 1,2 and 3 enclose sections of finer substrate. However,
we are unable to verify this as we have no camera footage or grab samples for ground-
Depression
Boundary 4 encloses depression and shadow. Again this is an educated guess due to
having no ground-
Sandy Substrate – Denser Seagrass
Boundary 7 contained a muddy substrate with varying densities of seagrass as can be seen on camera transect 2. This could be seen on the sidescan trace as spotty areas, which we can assume are patchy seagrass.
Sandy Substrate – Patchy Seagrass
The majority of the substrate around the features outlined is a sandy substrate with very patchy seagrass, as seen in camera transect 1. This is because the brightness around these areas are similar to those seen in the area of camera transect 1.
Rock – Kelp Forest
Boundaries 8 and 9 contain kelp and other marine flora and rocky substrate. This is based on camera transect 3, which shows these features.
Note
While there is a slight brightness change between EO184074m, NOO31612m and EO184073m,
NOO31479m, this line is not clearly defined and we have no ground-
Camera Transects – Results and Discussion
Camera Transect 1
Camera transect 1 showed fine sand and muddy sediments with sparse algae and seagrass (Zostera marina). No fauna are seen at the surface, but burrows and pits indicate the presence of infauna, such as burrowing polychaete worms.
Camera Transect 2
Camera transect 2 shows sand and muddy substrate with more abundant tufts of seagrass
(Zostera marina) and other algae. The presence of seagrass adds to information which
suggests that the sediment is sandy, as Z. marina grows in sandy sediment in subtidal
areas of 4 meters or less (Tyler-
Camera Transect 3
Camera transect 3 showed a shallow (4m depth) rocky habitat colonised by mixed vegetation including kelp (Laminaria species) and seaweed species, such as Saccorhiza polyschides and Heterosiphonia plumosa. H. pulmosa is a red seaweed which grows as an epiphyte on kelp species (Rowley, 2008a). The kelp from boundary sites 8 & 9 indicates several features associated with kelp growth. While kelp grows in most coastal areas with a temperate climate, most kelps require a hard substrate on which they can anchor and a high level of current. Currents cause sweeping which prevents sediment build up, and pushes away herbivorous organisms. The movement of canopies allows light to reach fronds which would otherwise remain in the shade. High current levels also allow for higher nutrients, particularly nitrate, as this is a very important requirement for kelp growth. (Dayton, 1985).
Camera transect 3 also shows ripple marks in the areas of sandy sediment covered by an algal carpet. These are characteristically formed in areas of constant high current, however these ripples are not big enough to be picked up by the sidescan sonar. Indications of infaunal organisms were also present.
Juvenile pollack (Pollachius pollachius) use estuarine algae as a nursery ground during their first year of life (Rowley, 2008b) and can be observed in the last still.
Budd, G., 2008. Asterias rubens. Common starfish. Marine Life Information Network [online] Available at: <www.marlin.ac.uk/speciesfullreview.php?speciesID=2657> [Accessed 28/06/2015]
Dayton, P. K., 1985. Ecology of Kelp communities. Annual Review of Ecology and Systematics.
16. pp. 215-
Deeble, M., Stone, V., 1985. A port that could threaten marine life in England’s
Fal Estuary. Oryx. 19:02 pp. 74-
JNCC, No Date A. Fal and Helford. [online] Available at: <jncc.defra.gov.uk/protectedsites/sacselection/sac.asp?EUcode=UK0013112> [Accessed 27/06/2015]
JNCC, No Date B. Special Areas of Conservation. [online] Available at: <jncc.defra.gov.uk/page-
JNCC, No Date C. Couch’s goby. [online] Available at: <jncc.defra.gov.uk/page-
Rowley, S., 2008a. Heterosiphonia plumosa. A red seaweed. [online] Available at: <www.marlin.ac.uk/speciesinformation.php?speciesID=3485> [Accessed 28/06/2015].
Rowley, S., 2008b. Pollachius pollachius. Pollack. [online] Available at: <www.marlin.ac.uk/speciesinformation.php?speciesID=4155> [Accessed 28/06/2015].
Somerfield, P. J., Gee, J. M., Warwick, R.M., 1994. Soft sediment meiofaunal community
structure in relation to long term heavy metal gradient. Marine Ecology Progress
Series. 105. pp. 79-
Tyler-

Camera still from camera 1

Camera still from camera 2

Camera still from camera 3

Camera still from camera 3
| Aims |
| Methods |
| Results and Discussion |
| References |
| Benthic Habitat Map |
| Cameras |
| Aims |
| Methods |
| Results |
| Discussion |
| Aims |
| Methods |
| Results |
| Discussion |
| References |
| Aims |
| Methods |
| Results and Discussion |
| References |
| Temperature and Density |
| Nutrients and Oxygen |
| Chlorophyll and Fluorescence |
| Plankton |
| ADCP |