|
Falmouth 2012 Group 11 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||


|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Alex Hewitt Cecilia Roos Foivi Kouraki Sally Stewart-Moore Sam Leach Sian Crowley Robin Rigby Roisin Quinn |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Top | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
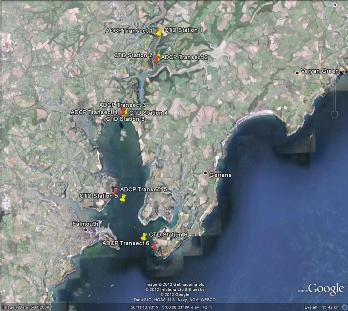
The Fal estuary (Figure 1.1) is the largest estuary in the UK, and the deepest in Europe (Pirrie et al 1995). It is a drowned river valley (ria) with a 127km shoreline, extending from the entrance (between Pendennis Point and St Anthony Head) 18km inland to the northern most tidal limit at Tresillian. It can be divided into two sections; the inner tidal tributaries and the outer tidal basin, known as the Carrick Roads which are characterised by a deep, narrow channel which ranges from 34m deep in the southern section to 5m in the River Fal (the northern section). The estuary is macrotidal at Falmouth with maximum spring tides of 5.3m and has a decreasing tidal amplitude towards Truro where maximum spring tides are 3.5m.
|
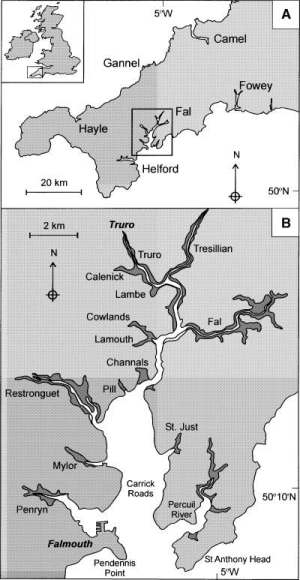 Figure 1.1: The Fal estuary system, located in Cornwall, southwest England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Top | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Boat Study
Introduction
The aim of our Fal estuary investigation was to study the spatial and temporal variations in stratification and to gain an understanding of the vertical and horizontal water column structure, the nutrient environment and the plankton community structure. Sample areas were chosen down the estuary (Figure 2.1) which provided insights into the physical, chemical and biological properties of the estuary. Temperature, salinity and light were measured to detect the physical properties of the estuary, while nutrient (dissolved silicate and phosphate) and dissolved oxyegn data provided an insight into the chemical properties of the estuary. Finally, the biological characteristics of the estuary were studied by measuring phytoplankton and zooplankton abundance.
In Situ Biological Methods
Table 1: Location of plankton net sampling
Plankton Net Plankton samples were taken at stations 1, 2 and 3 representing the upper estuary and 4, 5 and 6 representing the lower estuary. Each sample was collected using a plankton net (Figure 2.2) connected to a 1 litre bottle towed at constant speed and depth behind the boat. A proportion from this bottle was removed at each station and added to Lugols iodine solution; a preservative of phytoplankton. To the remaining water sample formalin was added; a preservative of zooplankton. The flow through the net was calculated using an impellor coupled to a five digit counter. This allowed for the variability of flow rate and thus plankton counts between stations to be accounted for.
In Situ Physical Methods
Table 2: A table to show location, depths and start times of each station
CTD
ADCP
Table 3: A table to show the location, depths and start time of the ADCP transect at each station
An Acoustic Doppler Current Profiler (ADCP) was used to measure the water current speed and direction at specific station along the estuary (Table 3).
In situ chemical methods
Phytoplankton
Zooplankton Zooplankton were analysed within a Bogorov chamber where abundance of each sample was recorded. Featured zooplankton included copepods, copepod nauplii, jellyfish larvae (cnidaria: hydrozoa), cirripedia, gastropod larvae and hydromedusa. Following this, the numbers recorded in each subsample were multiplied to provide an overall value of zooplankton abundance per metre cubed (Purdie, 2011). |
Figure 2.1: Map of CTD and ADCP station locations for the estuarine study
Figure 2.2: Phytoplankton net
Figure 2.3: CTD
Figure 2.4: Secchi disk
Figure 2.5: Samples bottles in the laboratory
Figure 2.6: Spectrometer |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Biological Results
A B C
D
Figures 2.7 (A-D): These pie charts show the composition of the phytoplankton species collected in the samples from the estuary station transects Figure 2.8: Bar chart showing number of cells per m3 of dinoflagellates, diatoms ciliates in transects A, B, C and D.
Phytoplankton Figure 2.8 shows the phytoplankton counts for Transects A-D in the estuary, and the phytoplankton composition of each sample. Overall, there was a general decline in phytoplankton abundance in a seaward direction from Transect A (5745 cells per m3) to Transect D (683 cells per m3), with the exception of the Transect C sample which at 7655 cells per m3 is higher than Transect B (2451 cells per m3). Diatoms dominated the phytoplankton in the samples from Transects A,C & D, while the Transect B sample was dominated by ciliates. Dinoflagellates formed a significant proportion of the phytoplankton from Transect A, although dinoflagellate representation declined downstream from A-D. The pie charts in Figure 2.7 show the relative composition of the phytoplankton from the water samples at Transects A-D. In Transect A, the phytoplankton was dominated by Coscinodiscus (Diatom) which accounted for 61.7% of the sample while the Transect B sample was dominated by Chaetoceros. Guinardia spp. (diatoms) formed a significant proportion of all the samples and were dominant in the samples from Transects C and D. Alexandrium was also relatively abundant in all the samples and Mesodinium rubrum formed a significant proportion of the phytoplankton in transect B. A B C D
Figures 2.9 (a-d)- These pie charts show the composition of the Zooplankton species collected in the samples from the estuary station transects Figure 2.10-Bar chart showing number of cells per m3 of zooplankton in transects A, B, C and D.
Zooplankton Figure 2.9(a-d) illustrate the composition of the zooplankton collected in the samples from transects A,B,C & D. Copepoda were the dominant zooplankton in all 4 transects, closely followed by Copepoda Nauplii. Other abundant groups throughout the transects included Hydromedusae, Cirripedia larvae and Decapoda larvae.
Biological Discussion
Phytoplankton The decline in phytoplankton abundance recorded with distance downstream can be attributed to a decline in nutrient concentration which ultimately limits growth. Furthermore, nutrient limitation is increasingly important during late Spring, when sampling occurred, due to nutrient depletion following the diatom spring bloom which occurs on an annual basis in temperate coastal waters (Sverdrup, 1953). This supports the evidence from Fig.2.8 which shows overall dominance by diatoms in the Fal estuary when sampling was carried out, while dinoflagellate abundance was relatively low, which indicates that diatoms tend to dominate the phytoplankton community during silicate sufficiency (Sommer, 1994). The decline in diatom dominance in a seawards direction may also reflect a decline in silicate concentration which diatoms have an ultimate growth requirement for (Tilman & Kilham, 1976). A long period of heavy rainfall prior to sampling may also be responsible for high phytoplankton abundance persisting for longer than normal.
Zooplankton
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chemical Results
Nutrients/Chlorophyll vs Depth Estuary
Station 1
Description Figure 2.11 Shows the changing concentrations of major nutrients and chlorophyll a with depth in the Fal estuary at station 1.
Discussion At station 1 high concentrations of phosphate are observed, due to low uptake from phytoplankton which can be seen from the low chlorophyll levels. The peak in chlorophyll corresponds to the minimum phosphate and dissolved silicon in the water column. High turbidity, centred around a null point of low flow in the estuary, may be contributing to the low concentrations of chlorophyll due to photoinhibition of phytoplankton growth and reproduction (Perianez, 2005; Garniera et al. 2010).
Station 2 Description Figure 2.12 shows the changing concentrations of major nutrients and chlorophyll a with depth in the Fal estuary at station 2.
Discussion At station 2 the concentration of phosphate begins to decrease, particularly in the mid water column, as the river water begins to mix with the phosphate-poor seawater. Chemical removal could also be contributing to the decrease in phosphate levels (Morris et al. 1981).
Station 3 Description Figure 2.13 shows the changing concentrations of major nutrients and chlorophyll a with depth in the Fal estuary at station 3.- detailed depth profile.
Discussion In surface waters here phosphate concentration decreases by ~300µmol L-1 from station 1 to station 3. At station 3, as well as there again being increased mixing between river water and seawater, the effect of phytoplankton uptake of phosphate from the water column begins to become apparent, as chlorophyll levels increase to a maximum of around 0.5 µg l-1 in the surface 2m.
Station 4 Description Figure 2.14 shows the changing concentrations of major nutrients and chlorophyll a with depth in the Fal estuary at station 4.- detailed depth profile: group 4 data.
Discussion At station 4 the chlorophyll levels increase significantly throughout most of the water column. These high chlorophyll levels correspond with the low nutrient concentrations, due to uptake by phytoplankton. From 5 to 9m chlorophyll levels rapidly decrease, which could be due to zooplankton consumption. The water column becomes much more homogeneous with regards to phosphate concentration at station 4.
Station 5 Description Figure 2.15 shows the changing concentrations of major nutrients and chlorophyll a with depth in the Fal estuary at station 5.- detailed depth profile: group 4 data.
Dissolved silicon concentration decreases slightly with depth from 3.9µmol/L in the surface waters to 2.7µmol/L at 12m.
Discussion Station 5 data shows an overall decrease in chlorophyll levels throughout the water column, but with a similar pattern of zooplankton grazing between 5 and 9m. There is also more pronounced evidence of zooplankton grazing in the top 2m. Chlorophyll levels may be lower overall due to a decrease in freshwater input with distance down the estuary. Phosphate levels remain very low, and dissolved silicon concentration also decreases to particularly low levels.
General Trends As can be seen from Figures 1 (riverine end member) to 5(estuarine end member) the phosphate concentration gradually decreases with proximity to the sea. This is due to an increase in mixing between seaward-flowing river water, which contains high concentrations of phosphate, and landward-flowing seawater, which contains much lower phosphate concentrations. As phosphate decreases significantly, chlorophyll levels increase, suggesting that the phytoplankton are utilising the phosphate. Chemical processes within the estuary may also be contributing to this removal. Concentration of dissolved silicon also decreases slightly throughout the estuary for similar reasons (Bien et al. 1958).
Estuarine Mixing
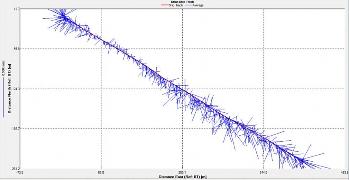
Dissolved silicon vs. Salinity
Description
Since sampling was not done further up
the estuary towards the riverine end, all data points are clustered
towards the marine end member. The lowest salinity reading was 27.3
and the highest was taken to be the marine end member (36.0).
Although all data points are within only a short distance from the
theoretical dilution line (TDL) there are points both above and
below the TDL with a greater weighting of points below the TDL. This
indicates mild removal of dissolved silicon from the estuary and
therefore non-conservative behaviour. Removal is most prevalent at
salinities between 27.3 (51.5µmol/L) and 35.0 (2.7µmol/L) which were
observed across all stations. Correlating this to phytoplankton
abundance from the plankton net trawls shows a dominance of diatoms
at all stations except B.
Discussion
Phosphate vs. Salinity
Description Again a wide range of salinity values towards the riverine end of the estuary were not sampled. Although all data points are based at the marine end member of the graph, they are widely varied in concentration values. The highest value (0.66 µmol/L) occurred at the most riverine sample (salinity 27.3) and the lowest value (0.09 µmol/L) at salinity 33.2. Overall a strong pattern of dissolved phosphate addition can be seen in waters of salinity range 27.3 to 35.0. However removal of phosphate can be seen at certain salinities (these include 33.2, 33.3, 34.2 and 34.9). The distance of data points above the TDL increases as salinity decreases indicating non-conservative behaviour.
Discussion
Dissolved phosphate is added to the
estuary. The degree of addition increases with distance up the
estuary (i.e. towards the riverine end). This addition, especially
to the upper reaches, can be attributed primarily to run-off from
the surrounding catchment area either directly or through
groundwater flow. This is exacerbated by the fact that the
freshwater catchment of the Fal lies in an extensive farming area
where intensive fertiliser agriculture and sewage-treatment is
practised (Langston et al., 2003). Furthermore the Fal catchment
area has underlying impermeable bedrock which along with high
rainfall experienced prior to and during the sampling periods
contributes vast concentrations of phosphate into the estuary (seen
in region of 27.3 and 35.0 salinity).
Physics
Salinity/temperature vs. Depth Station 1
Description Figure 2.18 shows that temperature undergoes a linear decrease from 14.5°C at the surface to 13.4°C at the maximum depth of 5m. Salinity shows an inverse relationship with temperature and undergoes a linear increase from a minimum 31 at the surface to a maximum of ~34 at 5m.
Station 2
Description Figure 2.19 illustrates a roughly inverse relationship between temperature and salinity collected by the CTD. Temperature rapidly decreases from 14.6°C to 13°C and salinity increases from 33 to 35, between the surface and 8m depth. Salinity remains fairly homogenous beyond 8 m, and temperature also stabilises somewhat at the same depth, only decreasing by around 0.2°C between 8 and 14 m.
Station 3
Description
Station 4 Description
Figure 2.21 shows a temperature decrease
as depth increases with surface temperatures of 14.3oC and
temperature at the bottom of the water column (12m) falling to just
below 12.0 oC. Greatest temperature decrease is seen between 2 and
4m depth, where temperature falls from 35oC to 33.3oC. As expected
there is an increase in salinity with increasing depth; surface
salinity of 32.8 and depth salinity just below 35.0 units. Greatest
salinity flux is at similar depths to greatest temperature flux,
with salinity increasing from 33.0 to 34.5 in approximately 1m of
water. The graph shows an inverse relationship between salinity and
temperature.
Station 5 Description Figure 2.22 also shows an inverse relationship between temperature and salinity. The greatest fluxes are seen between 2 and 4m. Temperature levels at the surface are at 14.3oC and fall to just below 13.0oC with greatest decrease seen in the upper 3m of the water column. Salinity levels at the surface are 33.8 and increase with depth to 35.0 at 20 m.
Station 6 Description Figure 2.23 showed much more variation in salinity and temperature. Furthermore the greatest flux of salinity and temperature is lower in the water column at station 6 and occurs at approximately 8 meters depth. The temperature level at the surface is 13.3 and falls to 12.6 at depth. Salinity fluctuates a lot more than temperature and there is an area of particularly low salinity at 2 meters depth where salinity has dropped from 34.9 to 34.7 however it then rises again until it reaches over 35.1 at 15 meters depth. At station 6 in the lower estuary there is an area of low salinity at the top of the water column (2m) which is likely to be a freshwater pocket sitting above the saline water.
General Discussion The general trend shown by the temperature salinity depth plots of the six stations shows temperature to decrease and salinity to increase with depth. The observed temperature decreases were due to solar radiation penetration which decreases with depth. Salinity increases with depth since saline water is denser than fresh water and thus sinks. Therefore the lighter freshwater flows out of the estuary above the denser saline water which flows into the estuary. This causes salinity stratification (halocline). A halocline can be seen in the upper section of the water column with the water below it being often homogenous or with just a small decrease in salinity. Across the 6 transects surface temperature remains fairly constant at around 14.5oC. However surface salinity varies greatly; in the upper estuary salinity is low (around 31.0) due to the high freshwater inputs from the many tributaries whereas in the lower estuary it is much higher (34.81) due to the mixing of saline waters. The Fal estuary is tidally dominated and impacted by freshwater inputs. Due to tidal mixing the change in temperature from surface to depth across the 6 stations is greater in the upper estuary, where mixing is less dominant, than in the lower estuary.
ADCP
Introduction
ρ2 is the density at the
deepest point,
Residence time
General Discussion Detailed description of results from stations 4 and 5 was not possible as there was a deep channel present half way through the transect making it impossible to collect measurements from the ADCP, therefore limited data were obtained. |
Figure 2.11- Chlorophyll, silicon and phosphate concentration with depth at station 1
Figure 2.12- Chlorophyll, silicon and phosphate concentration with depth at station 2
Figure 2.13- Chlorophyll, silicon and phosphate concentration with depth at station 3
Figure 2.14- Chlorophyll, silicon and phosphate concentration with depth at station 4
Figure 2.15- Chlorophyll, silicon and phosphate concentration with depth at station 5
2.16- Estuarine mixing diagram for dissolved silicon
Figure 2.17- Estuarine mixing diagram for dissolved phosphate
Figure 2.18- Temperature vs. Salinity profile for station 1
Figure 2.19- Temperature vs. Salinity profile for station 2
Figure 2.20- Temperature vs. Salinity profile for station 3 Figure 2.21- Temperature vs. Salinity profile for station 4 Figure 2.22- Temperature vs. Salinity profile for station 5 Figure 2.23- Temperature vs. Salinity profile for station 6
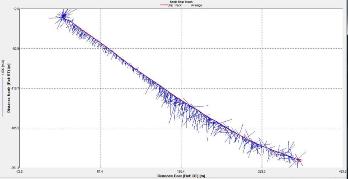
Figure 2.24: A depth profile to show how Ri number changes at station 1 though the water column.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
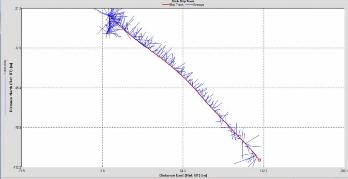
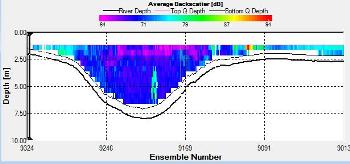
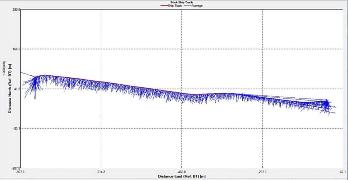
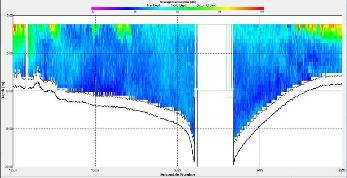
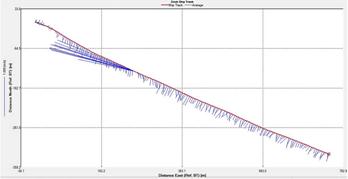
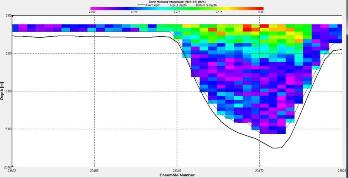
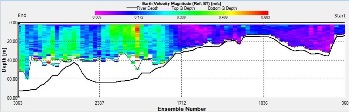
Figure 2.25: A ship stick track of the ADCP transect taken at station 1 at the top of the estuary (location 50° 14.390N, 05° 00.940W (at 08:32 UTC)) Figure 2.26: A velocity magnitude plot of the ADCP transect at station 1 Figure 2.27: A backscatter plot of the ADCP transect at station 1
Station 1
Looking at the velocity magnitude plot it can be seen that the lowest velocities are at the start (up to 2m) and end of the transect at surface and deep waters. The velocity decreases gradually from the surface to deeper waters moving from the end towards the start of the transect. The low velocities at the end of the transect come from an increase in shear stress which was created due to the change in the flow direction (see ship track). The highest velocities (red and orange boxes) are found at 4.89m with 0.31m/s, 1.89m with 0.36m/s and 1.39m with 0.31m/s. The greatest horizontal velocity difference occurred at 1.89m, with a change from 0.37m/s to 0.15m/s, showing a 60.0% change in velocity. The greatest vertical change in velocity was found between 4.39m and 3.89m with 0.40m/s and 0.18m/s respectively, showing a change of 54.3%. The Vertical water column is more stable and thus less susceptible to turbulence due to the effect of gravity and other stabilising factors, such as salinity and density limit spontaneous turbulence as more energy is required for mixing.
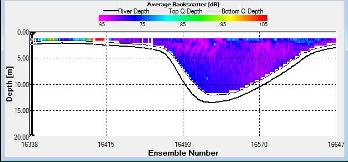
Figure 2.27 shows the highest biological or sediment activity at the start of the transect in the surface waters; 1.39m with 94dB. High backscatter patches (with an average of 76dB) were found in the middle of the transect, from 3- 6m, and towards the end of the transect where the high shear stress was present. |
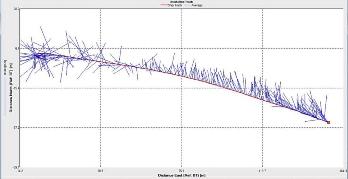
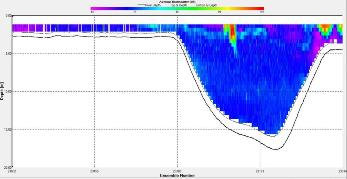
Figure 2.28 : The ship stick track for the ADCP at station 2 (location 50° 13.676N, 05° 01.030W at 09:27 UTC) Figure 2.29: A velocity magnitude plot of the ADCP transect at station 2
Figure 2.30: A backscatter plot of the ADCP transect at station 2
Station 2
The highest flow velocities were experienced at the middle and end of the transect, 0.39m/s at 11.39 m and an average of 0.35m/s around 4.39m. Towards the start of the transect flow velocities decreased. The greatest horizontal velocity change was seen at 4.89m with a 78.2% change (from 0.33m/s to 0.07m/s). The greatest vertical velocity change was observed between 4.89m and 5.39m, with 0.33m/s and 0.09m/s respectively, showing a 71.8% change.
The backscatter does not show any distinct patterns meaning there was not significant biological or sedimentary activity. The only high values (100dB and 105dB) were seen at the beginning of the transect in surface waters (1.39m) where there was increased turbidity due to shear stress experienced in shallow water.
|
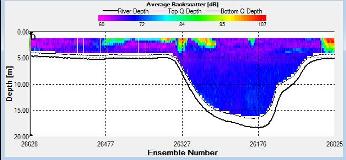
Figure 2.31 : The ship stick track for the ADCP at station 3 (location 50° 12.256 N, 05° 02.461 W, at 10:40 UTC) Figure 2.32: A velocity magnitude plot of the ADCP transect at station 3
Figure 2.33: A backscatter plot of the ADCP transect at station 3
Station 3 The flow is multidirectional throughout the whole transect with emphasis towards S and SW.
The velocity profile does not show any significant change in velocity. High velocities were seen at 11.39m (0.36m/s) at the start of the transect and towards the end of the transect at 4.39m (0.35m/s). The greatest horizontal change in velocity was found at 11.39m with a velocity change of 94.7% (from 0.36m/s to 0.02m/s). The greatest vertical change was observed between 4.39m and 3.89m; velocity change of 77.2% (from 0.35m/s to 0.08m/s).
Figure 2.33 shows a distinct area of high backscatter (107dB) at the start of the transect in surface waters decreasing down to 4m. A second distinct zone of high backscatter is seen at the sea surface in the middle of the transect; this is indicative of high biological activity most likely due to the presence of phytoplankton and/or zooplankton populations.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Figure 2.34 : The ship stick track for the ADCP at station 4 Figure 2.35: A velocity magnitude plot of the ADCP transect at station 4 Figure 2.36: A backscatter plot of the ADCP transect at station 4
Station 4 The direction of the flow (SW) is fairly constant throughout the transect. Just before gap in the middle of the transect (the depth of the water was greater than the depth that ADCP could read) the flow velocity experienced a great increase which can be seen in the much longer lines on the ship stick track in an easterly direction. Velocity also increased on the other side of the gap seen in the velocity magnitude plot as green and blue vertical line (average flow velocity of 2.6m/s). At other parts of the transect there was an overall average velocity of 0.3m/s. The greatest horizontal and vertical changes in velocity could not be found since vast amounts of data were missing which would have led to a loss of accuracy.
Strong backscatter is seen at the sea surface almost throughout the entire transect. The first section at the start of the transect has an average backscatter of 100dB and the section after the gap has an average of 85dB. The sections at the end of the transect have an average of 90dB. The strong backscatter is evidence for some strong biological activity. This could be a possible result of phyto- and/or zooplankton population activity.
|
Figure 2.37 : The ship stick track for the ADCP at station 5 Figure 2.38: A velocity magnitude plot of the ADCP transect at station 5 Figure 2.39: A backscatter plot of the ADCP transect at station 5
Station 5 The flow shows similar structure to the previous station. It is unidirectional for the whole transect apart from the deep point where flow shows an enormous increase. The overall direction of the flow was SW but changed when the deep point was reached to NW.
The velocity profile is fairly constant as was seen at the previous station. High velocities were observed at both sides of the gap. The principle for horizontal and vertical change is the same as at the previous station. |
Figure 2.40 : The ship stick track for the ADCP at station 6 Figure 2.41: A velocity magnitude plot of the ADCP transect at station 6
Figure 2.42: A backscatter plot of the ADCP transect at station 6
Station 6 Since this station was much shallower a full transect was able to be retrieved showing the separation of velocities from the surface waters within the channel and the bottom waters.
The flow is greatest in the surface waters of the channel (0.57m/s) and a minimum flow was seen at the bottom and sides of the channel (0.01m/s). Flow direction remains towards the mouth of the estuary since data was collected on the ebb tide. The flow is generally unidirectional (SW) throughout the first half of the transect. Over the second half spikes S and N moving away from the ship track are observed. At the sea surface the flow keeps the same direction showing laminar flow, however towards the deepest points, flow moves in varying directions indicating turbulence.
High velocities such as 0.57m/s are seen at the sea surface in the middle of the transect. At deeper layers the velocity is fairly constant with highest velocity (approximately 0.15m/s). The greatest horizontal velocity change was seen at 3.89m with 91.0% change (from 0.36m/s to 0.03m/s). The greatest vertical velocity change was observed between 3.39m and 3.89m, with a 76.45% change (from 0.31m/s to 0.07m/s). |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Pontoon Study
Introduction
The aim of the pontoon study was to gain an understanding of the temporal changes within the estuary over a tidal cycle. The water column was sampled every 15 minutes with data being recorded at 1m intervals. High tide occurred at 14:38 UTC (4.40m) which meant towards the end of the measurement time the water level dropped and measurements could only be taken down to 5m.
Physics
Salinity vs. Depth
Figure 2.43 shows time series data of salinity measured every 15 minutes off a pontoon from 9:00 UTC up to and including 17:45 UTC. It can be seen that salinity increases by 0.1 - 0.5 salinity units every 15 minutes up until 12:45. After 12:45 salinity begins to decrease steadily as time continues.
The initial increase in salinity coincides with high tide at 12:38 UTC, thus following the increase in marine water up into the estuary. The salinity decreases with time after high tide as the riverine water flows past the pontoon and out to sea. This is seen particularly clearly at the time closest to low water (9:00 UTC) which gave the lowest salinity value (surface 29.7 and three metres 32.2). Overall, there is a gradual increase (before high tide) followed by gradual decrease (after high tide) in salinity over time.
Flow Rate
Light Attenuation
Description
Discussion
Light Attenuation Coefficient (K)
In order to visualise how much suspended matter was being mixed throughout the water column along a time scale of a tidal cycle, the attenuation coefficient, K, was calculated and plotted on a time scale graph.
Description
Discussion
Chemistry
Description Figure 2.47 shows the change in Dissolved Oxygen measured over the tidal cycle from 09:00 – 17:45 (UTC) on the Pontoon. Throughout the sampling period, the concentration of dissolved oxygen showed an overall increase. Prior to high tide at 12:38 UTC, the concentration of dissolved O2 showed some fluctuations but increased gradually. No data was recorded immediately after high tide, and when sampling resumed at 14:45 there had been a marked decrease in dissolved O2. After 14:45, the concentration of dissolved O2 showed a steep and steady increase from 100.2µmol/l to 109.9µmol/l when sampling concluded at 17:45.
Discussion The results demonstrated an increase in dissolved oxygen concentration over time at the pontoon, with a sharper increase following high water. This change in dissolved oxygen can be associated with the increase in chlorophyll during the ebb tide caused by freshwater run-off, which leads to an increase in primary production by phytoplankton and thus an increase O2 production. Dissolved oxygen concentration is lower during the flood tide due to the progression up the estuary of sea water from the lower estuary which is depleted of oxygen.
Chlorophyll a Time Series
The chlorophyll a concentrations
increase with time as high tide is approached. Chlorophyll a
concentration is usually inversely correlated with tide stage when
inverse correlations occur between chlorophyll a and salinity (Welch
and Isaac, 1967).
|
Figure 2.43: A time series line graph to show how salinity varies with depth over time at a Pontoon located at 50°12.972N 05°01.659W.
Figure 2.44: Line plot showing the change in flow rates with depth at the pontoon. Line colour represents time (UTC) of sampling.
Figure 2.45: Line plot of the attenuation of light (Log10) in the water column over depth(m) measured at the pontoon. Line colour represents time of sampling (UTC).
Figure 2.46: Line graph illustrating the change in attenuation coefficient, k, over time on the pontoon.
Figure 2.47: Line graph illustrating the change in Dissolved Oxygen (µmol/l) over time on the Pontoon.
Figure 2.48: A time series line graph to show how chlorophyll a concentrations vary at a Pontoon located at 50°12.972N 05°01.659W. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Estuary Conclusion
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Top | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Introduction
The waters of the English Channel off Falmouth tend to be thermally stratified during the summer months and the strength of stratification is largely determined by a combination of water depth and tidal strength. However density structure, affected by freshwater inputs, wind strength and other climatic factors also influences stratification. As stratification stabilises the water column and thus affects vertical mixing, it has a profound effect on the physical and chemical properties of the surface layer of the ocean. In summer, surface waters in the English Channel tend to be nutrient depleted, while deep waters tend to be nutrient rich and therefore the rate at which growth limiting nutrients are mixed upwards in the thermocline is a key process determining biological productivity.
Table 4: A table to show the locations, depths and sampling times of each sampling station
Sampling was carried out on R.V. Callista on the morning of the 5th June 2012. Taking weather conditions into account, a transect extending out from the Fal estuary towards Lizard Point was designated, along which sampling stations were located. In order to locate the frontal system, sea surface temperature was monitored along the transect, and stations were chosen where significant changes in SST were observed.
The physical structure of the water column i.e. temperature and salinity at each station were measured using a CTD as for the estuarine sampling. Probes on the CTD Rosette system also measured turbidity, chlorophyll and irradiance.
Description can be found under the estuarine section of this website.
The ADCP was intended to be used to
describe the mixing regimes from the coastal to the offshore waters
off Falmouth. The boundary at which the waters changed from coastal
well mixed waters, to stratified offshore waters would give the
location of the front. This would then allow for a more informed
system of sampling either side of and at the boundary of the front.
The ADCP profiler could also be used in a biological manner to
locate the maxima of zooplankton due to the increase level of
backscatter at this point.
Chemical Methods CTD and Nutrients Data
Station 1- (figure 3.2)
In the upper 4m, temperature is constant
with depth, remaining at approximately 13.1°C. From 4-6m temperature
rapidly decreases from 13.1°C to 12.7°C and from 6-22m temperature
gradually decreases from 12.7°C to 12.6°C. In the surface 4m,
salinity is relatively constant with depth, remaining between
35.03-35.06. From 4-6m salinity rapidly increases from 35.05 to
35.15 and from 6-22m salinity gradually decreases from 35.15 to
35.19.
Discussion
At Station 1, the CTD profile for temperature and salinity can be attributed to the interaction of fresher, warmer estuarine water with cooler, more saline seawater at the mouth of the estuary. This profile may also have been enhanced by the ebbing tide at the time of sampling. This two-layered structure is typical of a partially mixed estuary in which tidal flow generates vertical mixing but is not strong enough to break down the difference in density between the two layers (Rockwell et al., 2000). The upper layer of less saline water may also be enhanced due to a period of high precipitation prior to sampling and there may also be a degree of surface warming from solar radiation, which is responsible for the higher temperatures at the surface.
Description
Discussion
The CTD depth profile for Station 2 represents a mixed water column, relative to the water column structure observed at Station 1. However there is a shallow fresher, warmer layer of water in the upper water column which is most likely a result of very high precipitation prior to sampling as well as solar energy warming the surface ocean. Below the upper few metres of the water column, salinity remains relatively uniform which indicates a significant amount of mixing. However temperature does decrease gradually with depth, and the notable decline in temperature at 30m may be due to wind mixing in the surface creating a layer of constant temperature down to the thermocline.
Description
Discussion Description
In the surface 5m, temperature decreases
rapidly from 14.2°C to 13.3°C. A rapid decrease in temperature with
depth can also be observed between 14m and 22m, where temperature
decreases from 13.2°C to 12.7°C. From 43-72m temperature remains
constant at 12.55°C. In the surface 5m, salinity varies greatly from
35.18 to 35.34. Below the varying surface waters, salinity remains
fairly uniform down the water column, remaining between 35.30 and
35.34. Discussion
The CTD profile for Station 4
demonstrates a stratified water column which can be associated with
the Frontal system we were searching for off Falmouth. This
stratified water column is comprised of 3 layers; a surface layer of
warmer, fresher water which again can be explained by freshwater
input and solar radiation; a middle layer with a marked reduction in
temperature and increase in salinity which are uniform down to 20m;
and a lower layer from 20m downwards which is characterised by
uniform high salinity, low temperatures and constant chlorophyll.
This provides a suitable example of seasonal thermal stratification
in the English Channel which creates a density gradient that
inhibits vertical mixing. On return to the estuary, the CTD was also deployed at station 5 in the deeper section of the channel. However the results from this profile represented a typical estuarine water column structure and therefore we chose not to analyse the data any further. |
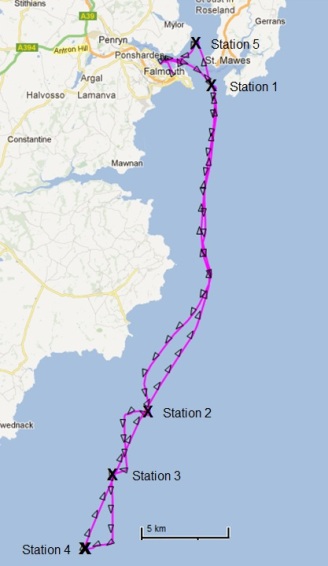 Figure 3.1 : A map of the plan A route and sampling stations along the route
Figure 3.2: CTD profile for Station 1. Red line representing change in temperature (°C) with depth and black line representing change in Salinity with depth. Blue line corresponds to the change in Chlorophyll (µg/l) with depth. Data points also show nutrient and dissolved oxygen concentrations (µmol/l) throughout the water column.
Figure 3.3: CTD profile for Station 2. Red line represents Temperature (°C) and Black line represents Salinity. Blue line corresponds to Chlorophyll (µg/l).
Figure 3.4: CTD profile for Station 3. Red line represents Temperature (°C) and Black line represents Salinity. Blue line corresponds to Chlorophyll (µg/l).
Figure 3.5: CTD profile for Station 1. Red line representing change in temperature (°C) with depth and black line representing change in Salinity with depth. Blue line corresponds to the change in Chlorophyll (µg/l) with depth. Data points also show nutrient and dissolved oxygen concentrations (µmol/l) throughout the water column. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Phytoplankton
Figure 3.6 (A & B): These pie charts show the composition of the phytoplankton species collected in the samples from the offshore station 1, depths 4.3 m (A) and 15.2 m (B).
Figure 3.7: These pie charts show the composition of the phytoplankton species collected in the samples from the offshore station 2, depth 31 m.
A B C Figure 3.8 (A-C): These pie charts show the composition of the phytoplankton species collected in the samples from the offshore station 4, depth 1.5 m (A), 17.9 m (B), 72.1 m (C.
Description
Guinardia flaccida dominated the sample from 4.3m at Station 1 in the estuary, accounting for 70.8% of the sample. Other species identified included Rhizosolenia setigera (20.8%) and Rhizosolenia stolterfothii (8.3%). At 15.2m (Figure 3.6), the sample was more diverse and was dominated by R. setigera (27.9%), closely followed by R. stolterfothii (22.7%). Other abundant species included Chaetoceros (18.2%) and G. flaccida (13.6%).
At Station 2 (Figure 3.7) the sample from 31m was also dominated by G. flaccida which accounted for 24.3% of the sample. Thalassiosira rotula (18.9%) and other unidentified Thalassiosira species (18.9%) also contributed to a significant proportion of the sample, as well as Coscinodiscus (10.8%) and Pseudo-nitschia (10.8%). At Station 4 (Figure 3.8) the 1.5m sample was also dominated by G. flaccida (31.2%), which was followed closely by Thallassiosira rotula (25.0%). R. setigera also formed a significant proportion of the sample, accounting for 18.8%, as well as Rhizosolenia alata which contriubted 15.6% of the sample. The sample from 17.9m was dominated by the species Leptocylindrus danicus which accounted for 48.3% of the sample. This was followed closely by Guinardia deliculata which formed 41.4%. Finally, at 72.1m, Chaetoceros and Karenia mikimotoi both formed a significant proportion of the sample, contributing 35.7% each.
Discussion
Zooplankton
Figure 3.9: Pie Chart representing the percentage composition of the zooplankton sample collected at Station 2 A Figure 3.10: Pie Charts representing the percentage composition of the zooplankton sample collected at Station 4 at (a) 0-10m and (b) 10-27m.
Description Copepoda dominated the zooplankton sample at Station 2, accounting for 63.1% of the sample, followed by Copepoda Naupilii which accounted for 10.0% of the sample. In the surface layer at station 4 (0-10m), the zooplankton was also dominated by Copepoda (40.3%) and followed by Cladocera which accounted for 22.3% of the sample. Appendicularia and Siphonophorae also formed a significant portion of the sample. In the lower water column at Station 4 (27-10m) the zooplankton community was more diverse although Copepoda were still the dominant group, forming 25.7% of the population. Other major groups included the Cladocera (18.6%), Appendicularia (14.3%), Chaetognatha (12.9%) and the Hydromedusae (10.0%).Discussion Copepoda dominated the zooplankton community at all the stations, which is typical for most cases. However, copepod dominance declined with distance offshore and other groups such as the Cladocera and Appendicularia became increasingly important. This transition in zooplankton community structure may be attributable to the frontal system located between stations 3 & 4, comprised of a thermally stratified water column in which phytoplankton are concentrated at the base of the euphotic zone. Therefore the zooplankton species observed in the deep sample may be grazing on the phytoplankton community above during the night time when they migrate vertically to the upper layer of the water column (Lampert, 1989).
ADCP From the graphs we can
see distinct patterns of thermocline along the water column. Several factors could have interfered with the accuracy of results for the calculation of the Ri number. As data were taken from both ADCP and CTD files, approximates of values were taken resulting in less accurate results. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
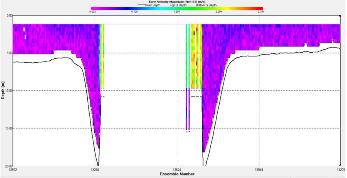
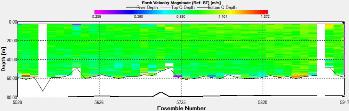
 Figure 3.11 : The ship stick track for the ADCP at station 1 Figure 3.12: A backscatter plot of the ADCP transect at station 4 Figure 3.13: A velocity magnitude plot of the ADCP transect at station 1
Station 1 At station 1 which was towards the estuary a distinct and strong thermocline from 4m to 6m can be seen; Ri number reached a maximum of 65, followed by mixed layers with Ri<1.
|
 Figure 3.14: A backscatter plot of the ADCP transect at station 4
Figure 3.15: A velocity magnitude plot of the ADCP transect at station 1
Station 2 At station 2, a logarithmic scale was used for the Ri number as it reached a value of 250. All the points at the right hand side of the depth line had Ri>1 and the ones on the left Ri<1; indicating layers of laminar and turbulent flow respectively.
|
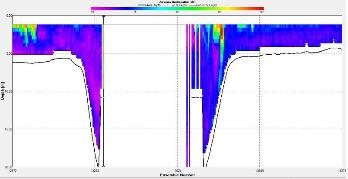
 Figure 3.16 : The ship stick track for the ADCP at station 3 Figure 3.17: A backscatter plot of the ADCP transect at station 4 Figure 3.18: A velocity magnitude plot of the ADCP transect at station 1
Station 3 At station 3, 1 weak and 2 strong thermoclines were apparent. The weak thermocline occurred between 14m and 20m, the first strong one between 30m and 36m and the second at 48m. The maximum Ri for the weak stratification was 2.33 at 14m and for the strong stratification was 35.5 at 32.5m for the first and 22 at 48m for the second. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Figure 3.19: A backscatter plot of the ADCP transect at station 4 Figure 3.20: A velocity magnitude plot of the ADCP transect at station 1
Station 4 At station 4 the same principle as for station 2 was applied due to relatively high values of Ri.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Offshore Conclusion
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Top | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Introduction
Geophysics Results
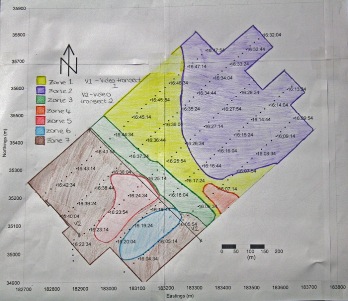
Zone 1
The fairly light shading indicates fine
sediment with no obvious bedforms present. A number of wakes can be
seen on the trace where turbulence caused small areas of data to be
unclear.
Most easterly area and the deepest part
of the scan. The trace indicates the sediment here is much finer,
seen by the light shading levels. Small seaweed streaks can be seen
as small dark dashes.
A clear channel is marked down the
centre of the trace, with the use of admiralty charts it can be
linked to a channel approaching Mylor harbour. This channel has been
dredged to 2.0m however at present it extends approximately 900m
from the harbour, most likely due to high boat traffic into the
harbour.
The smallest zone on the trace has been
outlined due to its darker shading in comparison to zone 1 which
encompasses it. There was no video data which could be linked to
this zone making it difficult to identify the change. The variation
in shading is likely due to a change to coarser sediment or possibly
more scallop beds.
The darkest shading occurred at zone 5.
Using the video data large areas of scallops and a small amount of
Maerl could be identified in this region. Furthermore very small
bedforms, due to shallow water influences, could be seen in the
northerly section of the zone.
The sidescan data at zone 6 indicated a
change in seabed material, and with the use of the video data from
the first video transect it can be verified that the change is due
to the presence of Maerl beds.
Using both the sidescan and images from
the drop camera on the first and second video transects, very dense
areas of seaweed were identified across the majority of zone 7. |
Figure 4.1: Transect lines plotted on computer.
Figure 4.2: Geophysics zoneation map |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Geophysics Conclusion
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Top | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
During the field course, the aim was to utilise the combination of three boat days (Offshore, Estuarine/Pontoon and Geophysical) in order to compare the Fal Estuary and the waters outside of the mouth. Data collection and processing through multi-parameter technique analysis allowed for comparison of the chemical, biological and physical characteristics of the environments.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Top | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Bien, G., Contois, D. and Thomas, W., 1958, "The Removal of Soluble Silica from Fresh Water Entering the Sea", Geochimica Et Cosmochimica Acta Volume 14.1, Issue 2, Pages 35-54
Cadee G.C. 1978, 'Primary production and chlorophyll in the Zaire River, Estuary and Plume, Netherlands', Journal of Sea Research, 12, 368-381
Cushing D.H. 1989, 'A difference in structure between ecosystems in strongly stratified waters and in those that are only weakly stratified.' Journal of Plankton Research, 11, 1-13. Garnier, J., Beusen, A., Thieu, V., Billen, G. and Bouwman, L., 2010, ‘N:P:Si nutrient export ratios and ecological consequences in coastal seasevaluated by the ICEP approach’, Global Biogeochem. Cycles, doi:10.1029/2009GB003583, in press.
GRASSHOFF, K., EHRDARDT, M., KREMLING, K. AND ANDERSON, L. G., 1999, ‘Methods of seawater analysis’, Wiley.
Irigoien X., Harris R.P., Head R.N. & Harbour D. 2000. 'North Atlantic Oscillation and spring bloom phytoplankton composition in the English Channel'. Journal of Plankton Research 22, 2637-2371
Lampert W. 1989. 'The adaptive significance of diel vertical migration of zooplankton.' Functional Ecology, 3, 21-27
Longston, W. J., Chesman, B. S., Burt, G. P., Hawkins, S. J., Readman, J. and Worsford, P. 2003, 'Characterisation of the south-west European marine sites: the Fal and Helford cSAC', Marine Biological Association, 8
Menesguen A. & Hoch T. 1997. Modelling the biogeochemical cycles of elements limiting primary production in the English Channel. I: Role of thermohaline stratification. Marine Ecology Progress Series 146: 173-188. Morris, A., Bale, A. and Howland, R., 1981, "Nutrient Distributions in an Estuary: Evidence of Chemical Precipitation of Dissolved Silicate and Phosphate", Estuarine, Coastal and Shelf Science Volume 12, Issue 2, Pages 205-16
Mortimer, R.J.G. et al. 1999, 'Sediment-Water Exchange of Nutrients in the Intertidal Zone of the Humber Estuary, UK', Marine Pollution Bulletin, 37, 261-279.
Mullin, J. B. and Riley, J. P.,1955, ‘The spectrophotometric determination of silicate-silicon in natural waters with special reference to sea water’, Anal Chim Acta, Volume 12, Pages 162-170
Parsons, T. R., Maita, Y. and Lalli, C. 1984, 'A manual of chemical and biological methods for seawater analysis', pergamon press, new york, 173
Pirrie et al., 1995, ‘Mineralogical and Geochemical Signiture of Mine Waste Contamination, Tresillian River, Fal Estuary, Cornwall, UK’, Environmental Geology, Volume 29, Ed:1-2, Pages 58-65
Rockwell G.W., Trowbridge J.H. & Bowen M.M. 2000. The Dynamics of a Partially Mixed Estuary. J. Phys. Oceanogr. (30): 2035–2048.
Somerfield, P. J., Gee, J. M. and Warwick, R. M., 1994, "Soft Sediment Meiofaunal Community Structure in Relation to aLong-term Heavy Metal Gradient in the Fal Estuary System", Marine Ecology Progress Series, Volume 105, Pages 79-88
Struyf E. et al., 2010, 'The global biogeochemical silicon cycle', Silicon, 1, 207-213
Welch, E. B. and Isaac, G. W. 1967,
'Chlorophyll variation with tide and with plankton productivity in
an estuary', 39, 360-366 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||